
Goldstein (1886) found that some rays passed through these holes in a direction opposite to that of the cathode rays. By using a discharge tube containing a perforated cathode. Hence after the discovery of the negatively charged constituent (electron) of an atom, attempts were made to discover the positively charged counterpart of electrons. “A sub−atomic particle which is a fundamental constituent of all matter having a mass 1/1837 th of a H−atom and which carries the smallest unit of negative charge is called an electron”.ĭISCOVERY OF PROTON: POSITIVE RAYS OR CANAL RAYSĪtoms are electrically neutral. Thomson named these negatively charged sub−atomic particles as electron. This indicates that the particles present in cathode rays are fundamental constituent of all matter. No cathode ray was produced when the tube was completely evacuated.ĭifferent gases produce same cathode rays as they have the same e/m (charge/mass) ratio. This shows that the particle is of sub−atomic nature.Ĭathode rays are deflected by a magnetic or an electric field showing the particle to be electrically charged, the direction of deflection shows that they are negatively charged.Ĭathode rays contain the smallest unit of negative charge. The mass of a particle present in cathode rays is found to be 1/1837 of H−atom. This shows that cathode rays contain material particles having both mass and velocity. They rotate a light wheel placed in their paths. They heat a substance on which they fall. The rays ionize a gas through which they pass. They produce X−ray when they strike a metal. If the metal sheet is too thick to be penetrated the rays cast a shadow. The rays pass through thin sheet of metals. They produce phosphorescence on certain salts like ZnS and fluorescence on glass. Their path is independent on the position of the anode. Thomson and others are given below:Ĭathode rays come out at right angles to the surface of the cathode and move in straight lines. Some of the important properties of the cathode rays studied by Sir J.J. When a current of high voltage (10,000 volts) is passed through a gas of air kept at a very low pressure (0.01 – 0.03 mm) blue rays are seen emerging from the case. The passage of electricity through gases as studied by a number of physicists, particularly by Faraday, Davy, Crookes and J.J. 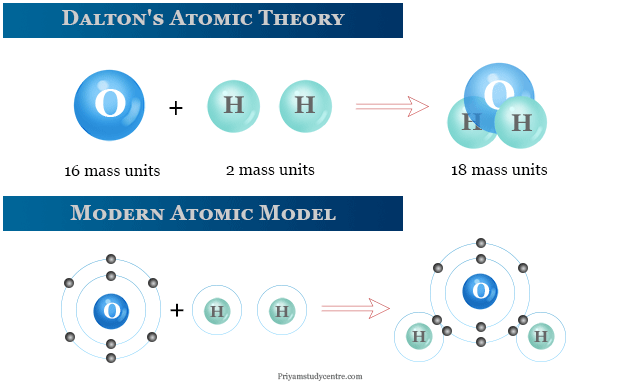
Though Dalton’s atomic theory could not give a convincing explanations to any of the above facts, it laid the foundation for the development of Modern atomic theory.The basic postulate of Dalton’s atomic theory which says that “Atom’s are the tiniest particles of matter which take part in Chemical reaction” is however accepted in modern atomic theory with experimental evidence.ĭuring the latter half of the nineteenth century, it was found that while normally dry gases do not conduct an electric current, they do so under very low pressure and then patches of light are seen. atoms of same elements may not be identical in all respect. the electron, proton, and neutron, it was concluded that atom can be further divided.ĭiscovery of isotopes proved that atoms of the same element may possess different atomic weights. ---teachoo-01-01.jpg)
With the discovery of subatomic particles, i.e. At present about 35 different subatomic particles are known but the three particles namely electron, proton and neutron are regarded as the fundamental particles.ĭalton’s atomic theory has been contradicted with the advancement of science and modified on the basis of further research and discoveries as follows: However, the researches done by various eminent scientists and the discovery of radioactivity have established beyond doubt that atom was not the smallest indivisible particle but has a complex structure of its own and was made up of still smaller particles like electrons, protons, neutrons etc. This means that a chemical reaction is just a simple rearrangement of atoms and the same number of atoms must be present before and after the reaction.Ītoms of different elements combine in a fixed ratio of small whole numbers to form compound atoms called molecules. Matter is made up of small indivisible particles called atom.Ītom is the smallest particle of an element, which takes part in a chemical reaction.Ītoms of the same element are identical in all respects especially in size, shape and mass.Ītoms of different elements have different mass, shape and size.Ītoms can neither be created nor be destroyed. The main postulates of Dalton’s atomic theory are: 
On the basis of large number of experiments performed, John Dalton proposed that matter was made up of extremely small particles called atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
